Search No Reviews Waking the Brain an Update on the Neurobiology
Traditionally, science has studied the human mind past observing how enquiry participants respond to external stimuli. In the grade of a mean solar day, however, we spend surprisingly little fourth dimension actively attention to stimuli in our immediate environment. First, during our waking hours, we spend about half of our time thinking virtually something other than our immediate surroundings—"heedless" or "mind-wandering" (Killingsworth and Gilbert, 2010). Beyond this, we spend nigh a 3rd of our lives sleeping. Yet the activeness of the brain, too as our accompanying stream of consciousness, persists throughout all these states of wakefulness and slumber. Here, several recent lines of show are described suggesting that dreaming and waking consciousness are not necessarily generated by independent mechanisms, running reverse to centuries of dogma on the fundamental nature of dreaming.
Conscious experience during sleep (i.eastward., dreaming) has classically been considered a phenomenon entirely singled-out from the spontaneous idea and imagery of wakefulness. But to the opposite, emerging testify suggests that dream experiences may best be conceptualized as a natural extension of waking consciousness, overlapping in both phenomenology and neural mechanism (Wamsley and Stickgold, 2010; Domhoff, 2011; Horikawa et al., 2013). In both resting wakefulness and sleep, the mind/brain is hard at piece of work processing the twenty-four hours's events and concerns—consolidating memory (Plihal and Built-in, 1997; Mednick et al., 2002; Tucker et al., 2006), integrating new information with our existing knowledge (Tamminen et al., 2010; Lewis and Durrant, 2011), and maybe fifty-fifty using by experience to plan for the future (Wilhelm et al., 2011). While dreaming and mind wandering are not necessarily functional in and of themselves, equally described beneath, emerging evidence suggests that these conscious experiences are influenced by the neurophysiological functions of the resting and sleeping brain. Thus, the systematic written report of subjective experience, across all states of consciousness, may prove crucial to a broader understanding of the brain function during "offline" states.
Toward a View of Dreaming equally a Normal Function of the Brain
Scientific progress in understanding dream consciousness has been woefully impeded past classical conceptions of dreams as a "mysterious" and "unknowable" miracle resistant to empirical investigation. This view is rooted in traditions that extend back thousands of years and nonetheless dominate popular conceptions of dreaming today. Even today, conscious experience during sleep is most ofttimes viewed equally originating in mechanisms split from those that generate normal waking thought and perception. In aboriginal cultures, this was expressed in the view that dreams originate outside the individual as divine messages from gods or spirits. In ancient Greece, for case, citizens suffering from physical ailments would flock to healing temples of the god Asclepius, where they would slumber and receive a divine dream that prescribed (upon estimation by a priest) a handling for their condition.
In the early on 20th century, the development of psychoanalytic dream theory was ostensibly a break from the superstitious traditions of the by, offering a scientific method of analyzing dreams from a psychological perspective. Even so this prototype nonetheless placed the origin of dreams in a mechanism exterior of the traditionally conceived "cocky"—now dreams came from the mysterious "unconscious mind," inaccessible during normal wakefulness and rife with sources of pathology. Calculation another layer of obscurity is the problematic notion that a dream experience can and should exist "interpreted." Despite thousands of years of dream interpretation, and the proliferation of dream symbol dictionaries on bookstore shelves, at that place is no systematic empirical evidence that dreams contain symbols to any greater caste than our typical waking thoughts, let lone has there been any empirical support for a particular system to "decode" these symbols. Although Freudian concepts of dreaming have at present fallen out of favor in many parts of the psychological customs, there has non been a widely accustomed new theory of dreaming to take its place.
Of class, information technology is non surprising that the question of dream consciousness received little scientific attention during the early-to-mid 20th century, when behaviorism dominated the mural of psychological research. Following the cognitive revolution, however, as psychologists and neuroscientists moved forrad with studying internal states such equally emotion, recollection, attention, and consciousness, there was little parallel boom of enquiry into subjective states during slumber (Figure 1). Thus, although cognitive neuroscientists take get increasingly comfortable with using introspective cocky-report to study wakefulness, conscious experience during dreaming has continued to be treated every bit a "special case" placed exterior the purview of scientific investigation.
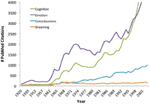
Effigy ane. The cerebral revolution has overlooked sleep. The cerebral revolution gear up off a research boom into a variety of long-neglected topics dealing with subjective experience during wakefulness, yet conscious feel during slumber and dreaming have non been targets of a comparable research surge. Counts represent the number of PubMed citations containing the keywords "cognition," "emotion," "consciousness" or "dreaming" (within all database fields and MeSH terms) for each year 1920–2003. The search for "dreaming" also included citations containing the MeSH term "dreams." Both enquiry articles and reviews are included in the commendation counts.
Even Alan Hobson's influential neurobiological theories of dreaming (Hobson and McCarley, 1977; Hobson et al., 2000) continued the historical thread of postulating a "special" machinery for dream generation, non-overlapping with those involved in waking mentation—In this instance, the machinery proposed was activity in the pontine brainstem occurring exclusively during the REM (rapid eye movement) phase of sleep. It is now articulate, however, that participants recall dreaming from l% or more than of awakenings outside of REM sleep likewise (Foulkes, 1962, 1967; Nielsen, 2000), including even during the deepest stages of wearisome wave slumber (SWS) (Cavallero et al., 1992; Cicogna et al., 2000). Thus, an eventual cognitive neuroscience of dream consciousness must invoke mechanisms that span across all of the classical "stages" of sleep.
Indeed, in the second one-half of the 20th century, several theorists began to draw dreaming as a variant of thought and imagery generation that spans us of wakefulness, REM, and NREM. The seminal work of Calvin Hall, for instance, was novel in presenting dream content as a relatively transparent reflection of a dreamers' waking life, rather than a mysterious psychological phenomenon specific to the slumber land (Hall, 1953). Afterward, the work of Antrobus (Antrobus, 1983, 1991; Reinsel et al., 1992) and Foulkes (1962, 1967) was cardinal in establishing that forms of circuitous, dreamlike mental activity occurred continuously throughout sleep onset, NREM sleep stages, and even resting wakefulness, suggesting dreaming as a signal on a continuum of forms of experience, rather than a phenomenon peculiar to ane slumber state. More contempo theories have continued to stress how the generation of dream consciousness is related to the neurobiology and cerebral structure of the waking encephalon (Cicogna and Bosinelli, 2001; Nir and Tononi, 2010; Wamsley and Stickgold, 2010; Perogamvros and Schwartz, 2012).
Several recent lines of testify continue to suggest that dream consciousness is generated by the same basic neural substrate that supports spontaneous subjective feel during "offline" states of resting wakefulness.
Comparing Conscious Experience in Resting Wakefulness and Slumber
One reason why dreaming has typically been treated separately from waking conscious experience is that there is causeless to be a bizarre, hallucinatory, and cognitively-scarce phenomenology of dreaming that clearly places it in a separate class of experience. But what is conscious experience really similar during sleep, and how does this differ from waking thought and imagery? Certainly, feel changes as we move through different states of consciousness—in comparison the form that experience takes during sleep, relative to wakefulness, perhaps the well-nigh noteworthy changes are an increase in the vividness of perceptual imagery coupled with attenuated awareness of the outside world (Hobson et al., 2000). Considering the desperate changes in neuromodulation, electrophysiology, and regional brain activation that accompany the onset of slumber, it is certainly not surprising that a corresponding shift in phenomenology would occur. Yet, despite articulate differences between waking heed-wandering and dreaming during slumber, there is little evidence to propose that conscious experience during dreaming is generated past a fundamentally different mechanism than during wakefulness. To the contrary, the available data suggest that dreaming during slumber overlaps in both phenomenology and neural mechanism with spontaneous mentation during offline wakefulness.
Debating whether dream consciousness and waking consciousness are "more than similar" or "more different" is a futile enterprise. It is clear that both differences and similarities be, but tabulating which listing has a greater number of items volition not necessarily allow us to draw any stiff conclusions. Nonetheless, it is important to annotation that the form and content of conscious experience in wake and sleep do overlap—subjective reports from these unlike states can, in fact, exist so similar as to be indistinguishable. Very vivid, fifty-fifty hallucinatory perceptual imagery, for example, is sometimes described in reports of waking daydreams (Foulkes and Scott, 1973; Foulkes and Fleisher, 1975). Meanwhile, dreams from sleep are not necessarily more "bizarre" than waking mentation. In fact, past ane measure, waking fantasy is more "bizarre" than dreaming—the number of sudden "discontinuous" shifts in topic is actually greater in reports of waking fantasy than in dreaming (Wollman and Antrobus, 1986; Reinsel et al., 1992). Conversely, cognition during slumber tin can be surprisingly logical and coherent, including self-reflection, planning, and focused attention (Kahan et al., 1997; Kahan and LaBerge, 2011).
Every bit an illustrative instance of the substantial overlap betwixt waking mentation reports and dreaming, below are two verbal reports from a single participant, one collected from resting wakefulness before sleep and 1 from Stage 2 NREM sleep:
I was picturing the dining room at my firm. Uh, it'due south kind of small because we take a very big tabular array in in that location; there'south about seven or 8 chairs around it and there's another big mirror on the wall, and it's blue – the room is blue. And, um, in that location's a smaller mirror with a gold frame to the left of the bigger mirror, and you can run into into the kitchen from the dining room. There's a little hallway that leads into information technology. (Resting Wake)
I was thinking most… I was in a room and there was someone from my Italian class there, just um, that's information technology… and there were tables and chairs in the room—kind of prepare similar desks, but that'south it. (Phase 2 NREM Sleep)
In REM sleep, dream experiences are oft longer, more than brilliant, and more "bizarre" than the examples higher up. Simply this is not necessarily the case. Although dream reports from REM are on average longer and more than detailed than those collected from NREM sleep, these distributions have substantial overlap (Foulkes, 1962, 1967; Antrobus et al., 1995; Cicogna et al., 1998; Smith et al., 2004; Wamsley et al., 2007). Importantly, many other apparent differences between REM and NREM dreaming (e.g., the amount of "baroque content," or the number of events and actions) can exist accounted for merely past their greater length (Antrobus, 1983).
Neurophysiological Correlates of Subjective Experience Across States of Consciousness
Dreaming has too been considered exterior the range of normal brain function because, past all outward appearances, the encephalon and mind are entirely "turned off" during slumber. Indeed, until the 1950's the predominant view of sleep was that of a global country of inactivity, where piddling or no brain and cognitive processing was occurring. The presence of complex thought and imagery was not easily reconciled with this classical view of the sleeping brain. However, post-obit the appearance of all-nighttime EEG recording, and more recently using PET and fMRI neuroimaging, nosotros can now see that the sleeping brain remains very active by several measures. The fast, desynchronized EEG of REM sleep, for example, so resembles that of waking brain action that this state was initially termed "paradoxical sleep." Even in the classically "deeper" stages of NREM sleep, neuroimaging studies prove that regional metabolic action is maintained in selected regions (Nofzinger et al., 2002; Peigneux et al., 2004).
Recent imaging studies have described a consequent pattern of brain activity present during resting wakefulness that overlaps substantially with activeness patterns during slumber [the "default network" (Buckner et al., 2008), encounter Figure 2]. Retentivity-related regions in the medial temporal and medial frontal regions are amongst the components of this that remain relatively active during both REM and NREM sleep (run across Domhoff, 2011 for a recent theoretical newspaper). During wakefulness, activation of the default network is associated with the generation of conscious thought and imagery (Stonemason et al., 2007; Andrews-Hanna et al., 2010; Andrews-Hanna, 2012). For example, default network activity is enhanced under conditions of reduced sensory monitoring that increment stimulus-independent thoughts (Andrews-Hanna et al., 2010). Furthermore, individuals reporting more task irrelevant thoughts of the past and futurity during a resting status exhibited increased functional connectivity between medial temporal lobe structures and other components of the default network (Andrews-Hanna et al., 2010). Finally, default network activation is also greater in individuals with a stiff trait propensity toward daydreaming (Mason et al., 2007). Thus, it appears that at that place is some structural and functional commonality between the "default manner" of resting wakefulness and patterns of preserved functional activation during sleep. The analogy, however, is non complete. First, parietal regions that grade a major component of the waking default network are relatively inactive during both REM and NREM slumber. Second, functional connectivity betwixt default network regions, which is a fundamental feature of how this network is divers during wakefulness, is altered as we enter sleep (Koike et al., 2011; Sämann et al., 2011).
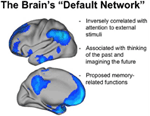
Figure 2. The "Default Network" of brain office. Functional imaging studies have identified a consistent network of encephalon regions that are preferentially active during periods of waking residuum, when participants are non engaged in processing external stimuli. Several of these aforementioned regions remain relatively active during sleep, including medial frontal and medial temporal networks involved in memory processing. (Adapted with permission from Buckner et al., 2008).
The relationship of default network activity to dreaming during sleep has non yet been straight tested. However, in that location is some preliminary evidence that particular regions of this network contribute to dream generation. For example, neuropsychologist Marking Solms has described lesions of the ventromedial prefrontal cortex which pb to a reported cessation of dreaming in affected patients (Solms, 2000). Likewise, recordings of intracranial EEG in epilepsy patients have demonstrated a relationship betwixt hippocampal activity during slumber and the recall of dream experience (Fell et al., 2006).
Thus, despite the credible inactivity of the sleeping body, and in the face of major neurophysiological changes, regional patterns of encephalon activity remain partially stable across the transition from resting wakefulness to slumber. During wakefulness, the co-activation of these encephalon regions is associated with the generation of thought, imagery, and heedless. If this functional network is similarly associated with conscious experiences arising during sleep, this would establish testify of a shared network for the generation of spontaneous subjective experience, which with some modification, spans across states of consciousness.
In Wake and Sleep, Spontaneous Conscious Feel Reflects Processing Past Memory and Planning for the Future
Cerebral neuroscience has now begun to study spontaneous experience during wakefulness in earnest. Emerging data show that far from beingness a meaningless distraction, so-called "daydreams" provide an important source of data about the activities of the resting brain. One line of this piece of work has been stimulated past involvement in the aforementioned "default network"—during periods of quiet residuum, activation of the default network (which includes several memory-related regions) is associated with remembering past events, but also with imagining possible future events (Addis et al., 2007; Andrews-Hanna et al., 2010; Andrews-Hanna, 2012). These observations have led to the hypothesis that one office of the brain during remainder is to use past memories in constructing simulations of possible futures, enhancing preparedness for futurity events (Schacter et al., 2007). Also in support of this hypothesis, patients with bilateral impairment to the hippocampus (a construction in the medial temporal lobe essential for forming new memories) bear witness not only memory impairments, but are besides dumb in their power to imagine fictitious scenarios and possible futures (Hassabis et al., 2007; Race et al., 2012). Together, with other evidence, these observations suggest that during periods of unoccupied residual, fragments of past feel are reactivated in our minds, and combined into novel imagined scenarios of possible futurity events.
Several lines of evidence suggest that dream experience may similarly reflect the processing of past retentivity, every bit well every bit the employ of memory to simulate future events. First, there is now very potent evidence that sleep is beneficial for the "consolidation" of newly acquired data. For both procedural (Stickgold et al., 2000a; Walker et al., 2002) and declarative (Plihal and Built-in, 1997; Ellenbogen et al., 2006; Tucker et al., 2006) forms of memory, mail service-learning slumber has consistently been shown to enhance later memory performance. Furthermore, the processing of retention during sleep appears to be direct reflected in the conscious feel of dreaming. Although by experiences are rarely, if ever, "replayed" in dreams in their complete and original course, nonetheless, a majority of dream reports contain at to the lowest degree one element which can be traced back to a specific recent memory (Fosse et al., 2003). Participants also very often dream most experimental learning tasks (Tauber et al., 1968; Stickgold et al., 2000b; Wamsley et al., 2010a, b; Kusse et al., 2011), and crucially, participants who incorporate learning tasks into their dream content evidence enhanced memory for the material post-obit sleep (Fiss et al., 1977; De Koninck et al., 1990; Wamsley et al., 2010b). Thus, although the content of dreams is unlikely to be exclusively determined by memory-related processes [for example, dreaming may also be influenced by motivational and reward systems (Pennartz et al., 2004; Perogamvros and Schwartz, 2012)], it appears that the consolidation of memory during sleep is one correspondent to the construction of dream feel.
Like waking daydreams, there is some preliminary bear witness that dreaming during sleep also reflects prospective memory functions, as the brain uses past experience to ready usa for the future. Beginning, we know that sleep does not enhance all past memories as, but instead selectively strengthens memory for information that is relevant to the future. For example, one contempo written report found that sleep only enhanced retentivity when participants expected to be tested the learned information the following morning time (Wilhelm et al., 2011). Similarly, sleep preferentially enhances emotional memories (Payne et al., 2008) and memories that participants expect to exist rewarded for remembering (Fischer and Born, 2009). Each of these studies illustrates a selective effect of slumber in enhancing retention for information that is important to an individual's future. At the aforementioned time, it has long been known that the simulation of potential futures forms a substantial role of dream content. For instance, Antti Revonsuo's "threat simulation" theory of dreaming builds on evidence that potentially threatening events are played out in imagined scenarios during dreams (Valli and Revonsuo, 2009). As another example, in our ain studies using the downhill skiing arcade game Alpine Racer Ii, we found that during a baseline recording dark, a minor merely meaning portion (4%) of dream reports anticipated playing Alpine Racer the following 24-hour interval, even though participants had never nevertheless seen the game. Taken together, these observations suggest the hypothesis that both spontaneous mentation during relaxed wakefulness and dreaming during slumber may be influenced past the same brain processes: the consolidation of past retention and effective simulation of future events.
Spontaneous Subjective Experience as a Tool for Cognitive Neuroscience
Studies of emotion, retentiveness, controlling, perception, and consciousness routinely rely on participants' own description of their internal states. Despite the unverifiable nature of such reports, progress in agreement human noesis has immensely benefitted from the employ of subjective report as a scientific tool. Why should dreaming exist treated any differently? Indeed, open-ended subjective reports from quiet rest and slumber were essential to much of the research described above. Self-report of ongoing conscious feel provides a method of determining whether a specific memory is existence reactivated in the resting encephalon (Wamsley et al., 2010a, b), and offers insight into other encephalon and cerebral processes which occurring during remainder (e.g., hereafter project, Andrews-Hanna, 2012) and sleep (east.g., reward processing, Perogamvros and Schwartz, 2012).
Importantly, there is as however no measure of brain activity (due east.yard., EEG, fMRI, PET) that can demonstrate the reactivation of a specific memory trace in the brain during man balance or sleep. For example, while an increase in hippocampal activity during slumber might indicate that memory processing is occurring (e.g., Peigneux et al., 2004), information technology cannot tell us whether a participant is reactivating the memory of a specific image, word, or thought. Emerging analytic techniques such as multivoxel pattern analysis of the fMRI Bold signal prove immense potential for decoding the neural correlates of recollecting a specific experience (Chadwick et al., 2010), but thus far, their application to defining offline memory reactivation remains in its infancy [though encounter (Horikawa et al., 2013)]. The witting retrieval of a contempo retentivity, in dissimilarity, conspicuously demonstrates that the neural networks encoding that detail memory take been reactivated. Thus, reports of conscious feel offer a valuable source of data about the activity of the resting brain, allowing us to make up one's mind which memories of everyday waking experience are spontaneously reactivated during offline states of quiet rest and sleep. Regardless of future progress in "decoding" experience based on encephalon signals, subjective study will continue to provide a valuable window into the cognitive processes occurring during offline states.
Of course, subjective study of experience during sleep does present some unique methodological challenges. Equally with all subjective report data, nosotros have access only to a participant'due south study of their recent feel, and no objective confirmation is bachelor [although meet (Horikawa et al., 2013)]. In dealing with reports of dream experience, this fundamental claiming is compounded by 2 boosted factors—Get-go, verbal reports of experience during sleep are necessarily given retrospectively, elicited only after a participant is awakened and entered a different land of consciousness. Second, memory for dream experiences is poor, relative to memory for waking feel. Yet, despite the quantitative reduction in call up of experience from sleep, there is no evidence that memory for sleep experiences is inherently less accurate than that for waking feel, and thus, there is no reason that such challenges should prevent us from utilizing these valuable data. Just as the challenges of subjective report accept not prevented progress in the report of emotion, retention, and consciousness, neither should the limitations of self-study prevent u.s. from studying subjective experience during slumber.
Dreams are not sent to us past the gods, nor are they a disguised message from the unconscious mind. Generated by the aforementioned mind and brain that create our waking conscious experience, dreams bear a transparent relationship to waking feel, and provide a useful window into activities of the sleeping brain. Because of this, introspective self-report is a valuable tool for the cognitive neurosciences. Moving into the future, the integration of behavioral, neural, and subjective data will enable us to map the structure, and potential function(s), of spontaneous thought across all states of consciousness.
Conflict of Interest Statement
The author declares that the research was conducted in the absence of any commercial or fiscal relationships that could exist construed as a potential disharmonize of interest.
Acknowledgments
This piece of work was conducted with support from Harvard Catalyst |The Harvard Clinical and Translational Scientific discipline Middle (National Center for Research Resource and the National Eye for Advancing Translational Sciences, National Institutes of Health Award 8UL1TR000170-05 and financial contributions from Harvard University and its affiliated bookish wellness care centers). The content is solely the responsibility of the authors and does not necessarily stand for the official views of Harvard Goad, Harvard Academy and its affiliated academic wellness care centers, or the National Institutes of Wellness.
References
Addis, D. R., Wong, A. T., and Schacter, D. Fifty. (2007). Remembering the past and imagining the future: common and distinct neural substrates during event construction and elaboration. Neuropsychologia 45, 1363–1377. doi: 10.1016/j.neuropsychologia.2006.x.016
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Andrews-Hanna, J. R., Reidler, J. S., Huang, C., and Buckner, R. Fifty. (2010). Testify for the default network's role in spontaneous cognition. J. Neurophysiol. 104, 322–335. doi: 10.1152/jn.00830.2009
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Antrobus, J., Kondo, T., Reinsel, R., and Fein, G. (1995). Dreaming in the belatedly morn: summation of REM and diurnal cortical activation. Conscious. Cogn. iv, 275–299. doi: 10.1006/ccog.1995.1039
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Buckner, R. Fifty., Andrews-Hanna, J. R., and Schacter, D. Fifty. (2008). The brain's default network: anatomy, function, and relevance to disease. Ann. N.Y. Acad. Sci. 1124, i–38. doi: 10.1196/register.1440.011
Pubmed Abstract | Pubmed Full Text | CrossRef Total Text
Cavallero, C., Cicogna, P., Natale, V., Occhionero, One thousand., and Zito, A. (1992). Slow moving ridge slumber dreaming. Slumber 15, 562–566.
Pubmed Abstract | Pubmed Full Text
Chadwick, G. J., Hassabis, D., Weiskopf, N., and Maguire, Due east. A. (2010). Decoding individual episodic memory traces in the homo hippocampus. Curr. Biol. xx, 544–547. doi: ten.1016/j.cub.2010.01.053
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Cicogna, P., Natale, V., Occhionero, Thou., and Bosinelli, Yard. (1998). A comparison of mental activity during sleep onset and forenoon awakening. Sleep 21, 462–470.
Pubmed Abstract | Pubmed Full Text
Cicogna, P., Natale, V., Occhionero, Yard., and Bosinelli, One thousand. (2000). Irksome wave and REM sleep mentation. Sleep Res. Online 3, 67–72.
Pubmed Abstract | Pubmed Full Text
De Koninck, J., Christ, 1000., Hébert, G., and Rinfret, N. (1990). Language learning efficiency, dreams and REM sleep. Psychiatr. J. Univ. Ott. 15, 91–92.
Pubmed Abstruse | Pubmed Full Text
Ellenbogen, J. One thousand., Hulbert, J. C., Stickgold, R., Dinges, D. F., and Thompson-Schill, Due south. L. (2006). Interfering with theories of sleep and memory: slumber, declarative retentiveness, and associative interference. Curr. Biol. xvi, 1290–1294. doi: 10.1016/j.cub.2006.05.024
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Fell, J., Fernández, G., Lutz, M. T., Kockelmann, Due east., Burr, W., Schaller, C., et al. (2006). Rhinal–hippocampal connectivity determines memory formation during sleep. Encephalon 129, 108. doi: 10.1093/encephalon/awh647
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Fiss, H., Kremer, E., and Litchman, J. (1977). The mnemonic function of dreaming. Sleep Res. 6, 122.
Foulkes, D. (1967). Nonrapid eye movement mentation. Exp. Neurol. (Suppl. iv), 28–38. doi: 10.1016/0014-4886(67)90154-9
CrossRef Full Text
Foulkes, D., and Scott, East. (1973). An above-zero baseline for the incidence of momentarily hallucinatory mentation. Sleep Res. 2, 108.
Hall, C. S. (1953). A cognitive theory of dreams. J. Gen. Psychol. 49, 273–282. doi: 10.1080/00221309.1953.9710091
CrossRef Total Text
Hassabis, D., Kumaran, D., Vann, S. D., and Maguire, E. A. (2007). Patients with hippocampal amnesia cannot imagine new experiences. Proc. Natl. Acad. Sci. U.Due south.A. 104, 1726–1731. doi: 10.1073/pnas.0610561104
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Hobson, J. A., and McCarley, R. Due west. (1977). The brain equally a dream country generator: an activation-synthesis hypothesis of the dream process. Am. J. Psychiatry 134, 1335–1348.
Pubmed Abstract | Pubmed Full Text
Hobson, J. A., Pace-Schott, Due east. F., and Stickgold, R. (2000). Dreaming and the encephalon: toward a cognitive neuroscience of conscious states. Behav. Brain Sci. 23, 793–842. word: 904–1121. doi: 10.1017/S0140525X00003976
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Kahan, T. Fifty., LaBerge, S., Levitan, L., and Zimbardo, P. (1997). Similarities and differences between dreaming and waking knowledge: an exploratory study. Conscious. Cogn. 6, 132–147. doi: 10.1006/ccog.1996.0274
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Koike, T., Kan, S., Misaki, M., and Miyauchi, South. (2011). Connectivity blueprint changes in default-manner network with deep non-REM and REM slumber. Neurosci. Res. 69, 322–330. doi: 10.1016/j.neures.2010.12.018
Pubmed Abstruse | Pubmed Full Text | CrossRef Total Text
Kusse, C., Shaffii-LE Bourdiec, A., Schrouff, J., Matarazzo, L., and Maquet, P. (2011). Experience-dependent induction of hypnagogic images during daytime naps: a combined behavioural and EEG written report. J. Slumber Res. 21, 10–twenty. doi: 10.1111/j.1365-2869.2011.00939.x
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Mason, Thou. F., Norton, M. I., Van Horn, J. D., Wegner, D. M., Grafton, Due south. T., and Macrae, C. Due north. (2007). Wandering minds: the default network and stimulus-contained thought. Science 315, 393–395. doi: 10.1126/science.1131295
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Mednick, Southward. C., Nakayama, K., Cantero, J. L., Atienza, M., Levin, A. A., Pathak, N., et al. (2002). The restorative consequence of naps on perceptual deterioration. Nat. Neurosci. five, 677–681.
Pubmed Abstract | Pubmed Total Text
Nielsen, T. A. (2000). A review of mentation in REM and NREM sleep: "covert" REM sleep as a possible reconciliation of two opposing models. Behav. Encephalon Sci. 23, 851–866. doi: 10.1017/S0140525X0000399X
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Nofzinger, Due east. A., Buysse, D. J., Miewald, J. M., Meltzer, C. C., Price, J. C., Sembrat, R. C., et al. (2002). Human regional cerebral glucose metabolism during non-rapid eye movement sleep in relation to waking. Brain 125, 1105–1115. doi: 10.1093/brain/awf103
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Payne, J. D., Stickgold, R., Swanberg, K., and Kensinger, E. A. (2008). Slumber preferentially enhances memory for emotional components of scenes. Psychol. Sci. 19, 781. doi: 10.1111/j.1467-9280.2008.02157.x
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Peigneux, P., Laureys, Southward., Fuchs, Southward., Collette, F., Perrin, F., Reggers, J., et al. (2004). Are spatial memories strengthened in the man hippocampus during deadening wave sleep? Neuron 44, 535–545. doi: 10.1016/j.neuron.2004.10.007
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Pennartz, C. Chiliad. A., Lee, Due east., Verheul, J., Lipa, P., Barnes, C. A., and McNaughton, B. L. (2004). The ventral striatum in off-line processing: ensemble reactivation during sleep and modulation by hippocampal ripples. J. Neurosci. 24:6446. doi: 10.1523/JNEUROSCI.0575-04.2004
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Race, E., Keane, M. M., and Verfaellie, One thousand. (2012). Losing sight of the futurity: impaired semantic prospection following medial temporal lobe lesions. Hippocampus 23, 268–277. doi: 10.1002/hipo.22084
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Reinsel, R., Antrobus, J., and Wollman, M. (1992). "Bizarreness in dreams and waking fantasy," in Neuropsychology of Sleep and Dreaming, eds J. Antrobus and Yard. Bertini (Hillsdale NJ: Lawrence Erlbaum Assembly), 157–184.
Pubmed Abstract | Pubmed Full Text
Sämann, P. G., Wehrle, R., Hoehn, D., Spoormaker, V. I., Peters, H., Tully, C., et al. (2011). Evolution of the brain'south default mode network from wakefulness to slow wave sleep. Cereb. Cortex 21, 2082–2093. doi: 10.1093/cercor/bhq295
Pubmed Abstract | Pubmed Full Text | CrossRef Total Text
Smith, Thou. R., Antrobus, J. S., Gordon, E., Tucker, M. A., Hirota, Y., Wamsley, E. J., et al. (2004). Motivation and affect in REM sleep and the mentation reporting process. Witting. Cogn. 13, 501–511. doi: x.1016/j.concog.2004.03.002
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Stickgold, R., Malia, A., Maguire, D., Roddenberry, D., and O'Connor, M. (2000b). Replaying the game: hypnagogic images in normals and amnesics. Scientific discipline 290, 350–353.doi: ten.1126/Science.290.5490.350
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Tamminen, J., Payne, J. D., Stickgold, R., Wamsley, E. J., and Gaskell, 1000. G. (2010). Sleep spindle activity is associated with the integration of new memories and existing knowledge. J. Neurosci. 30, 14356–14360. doi: 10.1523/JNEUROSCI.3028-x.2010
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Tauber, E. S., Roffwarg, H. P., and Herman, J. (1968). The effects of longstanding perceptual alterations on the hallucinatory content of dreams. Psychophysiology 5, 219.
Tucker, Yard. A., Hirota, Y., Wamsley, E. J., Lau, H., Chaklader, A., and Fishbein, W. (2006). A daytime nap containing solely not-REM sleep enhances declarative but non procedural memory. Neurobiol. Learn. Mem. 86, 241–247. doi: 10.1016/j.nlm.2006.03.005
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Valli, One thousand., and Revonsuo, A. (2009). The threat simulation theory in light of recent empirical bear witness: a review. Am. J. Psychol. 122, 17–38.
Pubmed Abstract | Pubmed Total Text
Walker, Thou. P., Brakefield, T., Morgan, A., Hobson, J. A., and Stickgold, R. (2002). Do with sleep makes perfect: sleep-dependent motor skill learning. Neuron 35, 205–211. doi: 10.1016/S0896-6273(02)00746-eight
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Wamsley, Eastward. J., Hirota, Y., Tucker, M. A., Smith, M. R., and Antrobus, J. S. (2007). Cyclic and ultradian influences on dreaming: a dual rhythm model. Brain Res. Balderdash. 71, 347–354. doi: 10.1016/j.brainresbull.2006.09.021
Pubmed Abstract | Pubmed Full Text | CrossRef Total Text
Wamsley, East. J., Perry, Thousand., Djonlagic, I., Reaven, 50. B., and Stickgold, R. (2010a). Cerebral replay of visuomotor learning at sleep onset: temporal dynamics and relationship to chore functioning. Sleep 33, 59–68.
Pubmed Abstract | Pubmed Full Text
Wamsley, E. J., Tucker, G., Payne, J. D., Benavides, J. A., and Stickgold, R. (2010b). Dreaming of a learning task is associated with enhanced sleep-dependent memory consolidation. Curr. Biol. 20, 850–855. doi: x.1016/j.cub.2010.03.027
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Wilhelm, I., Diekelmann, S., Molzow, I., Ayoub, A., Mölle, M., and Born, J. (2011). Slumber selectively enhances memory expected to exist of future relevance. J. Neurosci. 31, 1563–1569. doi: x.1523/JNEUROSCI.3575-10.2011
Pubmed Abstruse | Pubmed Full Text | CrossRef Total Text
Wollman, M. C., and Antrobus, J. South. (1986). Sleeping and waking thought: effects of external stimulation. Sleep 9, 438–448.
Pubmed Abstract | Pubmed Full Text
Source: https://www.frontiersin.org/articles/10.3389/fpsyg.2013.00637/full
0 Response to "Search No Reviews Waking the Brain an Update on the Neurobiology"
Post a Comment